In Search of Memory: The Emergence of a New Science of Mind (12 page)
Read In Search of Memory: The Emergence of a New Science of Mind Online
Authors: Eric R. Kandel
Tags: #Psychology, #Cognitive Psychology & Cognition, #Cognitive Psychology

What accounts for this difference in voltage? Bernstein reasoned that something must carry electrical charge across the cell membrane. He knew that every cell in the body is bathed in the extracellular fluid. This fluid does not contain free electrons to carry current, as metal conductors do; instead it is rich in ions—electrically charged atoms such as sodium, potassium, and chloride. Moreover, the cytoplasm inside each cell also contains high concentrations of ions. These ions could carry current, Bernstein reasoned. He had the further insight that an imbalance in the concentration of ions inside and outside the cell could give rise to current across the membrane.
Bernstein knew from earlier studies that the extracellular fluid is salty: it contains a high concentration of sodium ions, which are positively charged, balanced by an equally high concentration of chloride ions, which are negatively charged. In contrast, the cytoplasm of the cell contains a high concentration of proteins, which are negatively charged, balanced by potassium ions, which are positively charged. Thus the positive and negative charges of ions on either side of the cell membrane are balanced, but different ions are involved.
For electrical charge to flow through the nerve cell membrane, the membrane must be permeable to some ions in the extracellular fluid or the cytoplasm. But which ions? After experimenting with various possibilities, Bernstein arrived at the bold conclusion that in the resting state, the cell membrane presents a barrier to all ions except one—potassium. The cell membrane, he argued, contains special openings, now known as ion channels; these channels allow potassium ions, and only potassium ions, to flow along a concentration gradient from the inside of the cell, where they are present in high concentrations to the outside, where they are present in low concentrations. Since potassium is a positively charged ion, its movement out of the cell leaves the inside surface of the membrane with a slight excess of negative charge resulting from the proteins inside the cell.
Even as potassium moves out of the cell, however, it is drawn back toward the inside of the cell by the net negative charge it leaves behind. Thus the outside surface of the cell membrane becomes lined with positive charges from the potassium ions that have diffused out of the cell, and the inside of the membrane becomes lined with negative charges from the proteins attempting to draw potassium ions back into the cell. This balance of ions maintains the stable membrane potential of–70 millivolts (figure 5–3).
These fundamental discoveries about how nerve cells maintain their resting membrane potential led Bernstein to ask: What happens when a neuron is stimulated sufficiently to generate an action potential? He applied electrical current from a battery-operated stimulator to a nerve cell axon to generate an action potential and inferred that the selective permeability of the cell membrane breaks down very briefly during the action potential, allowing all ions to enter and leave the cell freely and reducing the resting membrane potential to zero. According to this reasoning, by moving the cell membrane from its resting potential of–70 millivolts to 0 millivolts, the action potential should be 70 millivolts in amplitude.
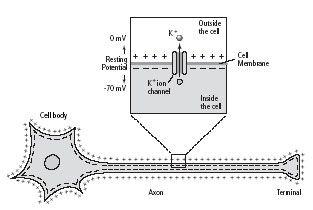
5–3 Bernstein’s discovery of the resting membrane potential.
Julius Bernstein deduced that there is a difference in voltage between the inside and the outside of a nerve cell, even in its resting state. He proposed that the nerve cell membrane must have a special channel through which positively charged potassium ions (K+) can leak out of the cell and that this loss of positive charge leaves the inside surface of the cell membrane negatively charged and creates the resting membrane potential.
The membrane hypothesis formulated by Bernstein proved powerful, in part because it was based on well-established principles of ions moving in solution and in part because it was so elegant. The resting potential and the action potential did not require elaborate biochemical reactions but simply utilized the energy stored in the concentration gradients of the ions. In a larger sense, Bernstein’s formulation joined those of Galvani and Helmholtz in providing compelling evidence that the laws of physics and chemistry can explain even some aspects of how mind functions—the signaling of the nervous system and therefore the control of behavior. There was no need or room for “vital forces” or other phenomena that could not be explained in terms of physics and chemistry.
THE FOURTH PHASE WAS DOMINATED BY THE IONIC HYPOTHESIS
and by the thinking of Alan Hodgkin, Adrian’s most brilliant student, and Andrew Huxley, Hodgkin’s own talented student and colleague (figure 5–4). Hodgkin and Huxley’s working relationship was collaborative and synergistic. Hodgkin had piercing biological and historical insights into how nerve cells function. A fine experimentalist and a superb theoretician, he was always looking for the larger meaning beyond the immediate finding. Huxley was a technically gifted mathematical whiz. He devised new ways of recording and visualizing the activity of single cells, and he developed mathematical models to describe the data he and Hodgkin obtained. They were perfect collaborators, more than the sum of their parts.

5–4
Alan Hodgkin (1914–1998) and Andrew Huxley (b. 1917) carried out a series of classic studies in the giant axon of squid nerve cells. Besides confirming Bernstein’s notion that the resting membrane potential is caused by the movement of potassium ions out of the cell, they discovered that the action potential is caused by the movement of sodium ions into the cell. (Courtesy of Jonathan Hodgkin and A. Huxley.)
Hodgkin’s enormous gifts were evident early in his career, and by the time he started his collaboration with Huxley in 1939, he had already made a major contribution to neuronal signaling. In 1936, he won a research fellowship for Trinity College, University of Cambridge, in England based on his dissertation on the “Nature of Conduction in Nerve.” There he showed, in elegant quantitative detail, that the current generated by an action potential is large enough to jump over an anesthetized segment of the axon and prompt the unanesthetized portion beyond it to generate an action potential. These experiments provided the final insights into how action potentials, once initiated, can propagate without failure or flagging. They do so, Hodgkin demonstrated, because the current generated by the action potential is substantially greater than the current required to excite a neighboring region.
The research described in Hodgkin’s dissertation was so important and so beautifully executed that it immediately brought him, at the age of twenty-two, to the attention of the international scientific community. A. V. Hill, a Nobel laureate and one of England’s leading physiologists, sat on Hodgkin’s dissertation committee and was so impressed that he sent the dissertation to Herbert Gasser, president of the Rockefeller Institute. In his covering letter, Hill referred to Hodgkin as “very remarkable…. It is almost an unheard-of thing for an experimental scientist at Trinity College in Cambridge to get a fellowship in his fourth year, but this youngster has done it.”
Gasser found young Hodgkin’s dissertation to be a “beautiful job of experimentation” and invited him to spend 1937 as a visiting scientist at Rockefeller. During that year Hodgkin befriended Grundfest, who worked in the laboratory next door. Hodgkin also visited a number of other laboratories in the United States and in so doing learned about the squid’s giant axon, which he would subsequently use to great advantage. Finally, he met the woman he would ultimately marry, the daughter of a Rockefeller Institute professor. Not a small set of accomplishments for just one year!
Hodgkin and Huxley’s first great insight came in 1939, when they traveled to the marine biological station at Plymouth, England, to study how the action potential is generated in the giant axon of the squid. The British neuroanatomist J. Z. Young had recently discovered that the squid, one of the fastest swimmers in the sea, has a giant axon that is a full millimeter (1/25 of an inch) in diameter, making it about a thousand times wider than most axons in the human body. It is about the width of a piece of thin spaghetti and can be seen with the naked eye. Young, a comparative biologist, understood that animals evolve specializations that enable them to survive more effectively in their environments, and he realized that the squid’s specialized axon, which powers its rapid escape from predators, could prove a godsend for biologists.
Hodgkin and Huxley sensed immediately that the giant axon of the squid might be just what they needed to pursue the neural scientist’s dream of recording the action potential from inside the cell as well as from outside it, thereby revealing how the action potential is generated. Because that axon is so large, they could thread one electrode into the cytoplasm of the cell while keeping another on the outside. Their recordings confirmed Bernstein’s inference that the resting membrane potential is about–70 millivolts and that it depends on the movement of potassium ions through ion channels. But when they stimulated the axon electrically to produce an action potential, as Bernstein had done, they discovered to their amazement that it was 110 millivolts in amplitude, not the 70 millivolts Bernstein had predicted. The action potential had increased the electrical potential at the cell membrane from–70 millivolts at rest to +40 millivolts at its peak. This astonishing discrepancy had a profound implication: Bernstein’s hypothesis that the action potential represents a generalized breakdown of the cell membrane’s permeability to all ions had to be incorrect. Rather, the membrane must still be acting selectively during the action potential, allowing some ions but not others to move across it.
This was an extraordinary insight. Since action potentials are the key signals for conveying information about sensations, thoughts, emotions, and memory from one region of the brain to another, the question of how the action potential is generated became, in 1939, the dominant question in all of brain science. Hodgkin and Huxley thought deeply about it, but before they could test any of their ideas, World War II intervened and both were called into military service.
It was not until 1945 that the two men could return to their research on the action potential. Working briefly with Bernard Katz of University College, London (while Huxley was preparing to get married), Hodgkin discovered that the upstroke—the rise and eventual height of the action potential—depends on the amount of sodium in the extracellular fluid. The downstroke—the decline of the action potential—is affected by the concentration of potassium. This finding suggested to them that some ion channels in the cell are selectively permeable to sodium and are open only during the upstroke of an action potential, whereas other ion channels are open only during the downstroke.
To test this idea more directly, Hodgkin, Huxley, and Katz applied a voltage clamp, a newly developed technique for measuring ion currents across the cell membrane, to the squid’s giant axon. They again confirmed Bernstein’s finding that the resting potential is created by the unequal distribution of potassium ions on either side of the cell membrane. Moreover, they confirmed their earlier finding that when the cell membrane is sufficiently stimulated, sodium ions move into the cell for about 1/1000 of a second, changing the internal voltage from–70 millivolts to +40 millivolts and producing the rise of the action potential. The increased sodium inflow is followed almost immediately by a dramatic increase in potassium outflow, which produces the decline of the action potential and returns the voltage inside the cell to its initial value.
How does the cell membrane regulate the change in sodium and potassium ion permeabilities? Hodgkin and Huxley postulated the existence of a previously unimagined class of ion channels, channels with hinged “doors,” or “gates,” that open and close. They proposed that as an action potential propagates along an axon, the gates of the sodium and then the potassium channels open and close in rapid succession. Hodgkin and Huxley also realized that, because the opening and closing of the gates is very fast, the gating must be regulated by the voltage difference across the cell membrane. They therefore referred to these sodium and potassium channels as
voltage-gated channels
. In contrast, they called the potassium channels that Bernstein had discovered and that are responsible for the resting membrane potential
non-gated potassium channels
, because they have no gates and are unaffected by the voltage across the cell membrane.
